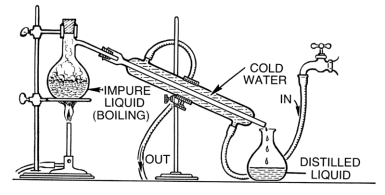
Ethanol Alcohol, in its pure form, is safe to drink and can be used in various experiments. However, in order to discourage its abuse, denaturing substances such as benzene and arsenic are added, therefore making it toxic and harmful to drink. In order to obtain it again, the denatured version which comes as an ingredient in different products, needs to be distilled.

What you will Need
- Balance Scale
- 100-ml Volumetric Flask or Graduated Cylinder
- Distillation Apparatus
- 250-ml Beaker for receiving the Distilled Ethanol
- Hotplate or any other heatless source
- Boiling Chips
- 200-ml Denatured Ethanol
Directions
- Before anything else, make sure to put protective gear such as goggles, gloves and protective clothing.
- Add 100 ml ethanol to the volumetric flask then weigh it. After that, pour it inside the distillation vessel along with the remaining ethanol.
- Add a boiling chip to the flask before assembling the distillation apparatus.
- Turn on the hot plate and heat the alcohol to a gentle boil while measuring its temperature at the same time. Make sure that the temperature is only stable when you’re heating it.
- After distilling it, let it cool under room temperature.
- If you want it to be more pure, you can distill it again.
If you’re looking for denatured ethanol, it usually comes in form of ethyl alcohol or ethyl rubbing alcohol and is usually sold in local pharmacies. On the other hand, isopropyl alcohol is also being sold as a rubbing alcohol, too so be careful in reading the bottle’s labels before doing anything.